Corrosion is one of the most common reason for failures, and the additional expenses due to it are counted in billions. Especially, in electrical applications corrosion can very quickly destroy the functionality of devices and in worst case be a major safety risk. Consequently, it is critical to know how to protect your products against corrosion and its effects.
Considering the importance of corrosion, it is not surprising that there is a lot of interest to find ways to determine how to protect products against corrosion. However, analysing the reasons for corrosion is not simple. The problem is that there are so many factors which affect corrosion. Some of them depend on the product itself. For example, some materials contain corrosive impurities which can cause problems in suitable conditions. However, many factors affecting corrosion depend on the environment the product is used or stored in. For example, air pollution or industrial exhausts can have significant effect on corrosion.
If you want to understand how corrosive your use and storage environments are, it would be important to measure them. Unfortunately, measuring corrosivity is not simple. Some environmental factors are quite easy to measure. For example, both humidity and temperature are critical factors for corrosion. In an excellent overview of corrosion by VTT, it is stated that relative humidity level of 40% is needed for corrosion to occur. However, if temperature reaches 40°C, corrosion can occur even with low humidity levels. Both humidity and temperature are relatively easy to measure using the numerous sensors available.
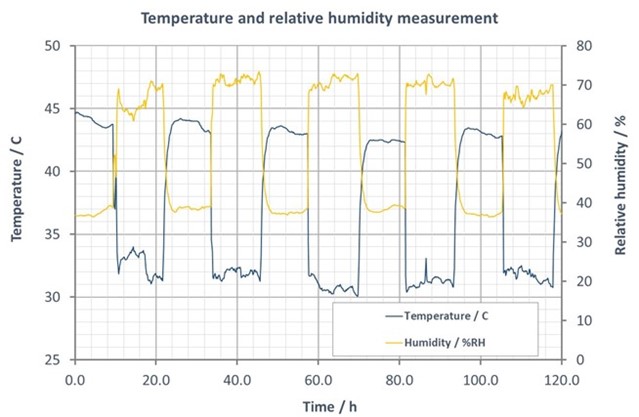
Relative humidity and temperature typically vary between day and night in outdoor conditions.
Unfortunately, most corrosive elements are far more difficult to measure. Typically, complicated and expensive measurement systems are needed to get proper results. Furthermore, corrosive elements may mask each other in measurements, making their analysis even more complicated. Moreover, you need to know what you are measuring to optimize the measurements for these elements. This might not be easy to determine.
Corrosion coupons have been used for a long time to analyse the corrosiveness of environments. The idea is to use metallic coupon and expose it to the studied environment for a predetermined time period, for example a month. After exposure the amount of corrosion on the coupon is analysed and this information is used to assess the corrosivity level of the environment. There are several standards, for example ISO 9223:2012 and ISA S71.04-2013 which determine corrosiveness levels and help to understand how corrosive your use environments really are.

Copper corrosion coupons are commonly used to analyse the corrosiveness of environments.
The main drawback of corrosion coupons is that they do not enable in-situ monitoring and in order to get results you have to wait for a month or even more. However, there greatest benefit is that the coupon reacts to all corrosive elements in the environment. By using coupons made of different metals you can get quite easily get a good approximation of how corrosive the environment is. Coupons can be analysed using various techniques. In our corrosion monitoring device, we use weighing and coulometric reduction and occasionally also mass reduction analysis. The benefit of coulometric reduction is that it gives you information of the corrosion products and this information can give more information of the environment.

Trelic’s Corrosion Monitoring Cube can be used to study corrosiveness of many different environments.
So why should you measure the corrosiveness of environments? As stated in the beginning, corrosion is one of the most common reasons for failures which causes huge additional costs for companies. Early failures of products are also against the aim for more sustainable products. By analysing the use and storage environments you can understand the level of risk due to corrosion. This helps to determine how much protective measures are needed. Additionally, you can use this information in corrosion test design and to find the right test methods for your product. The correlation between the test conditions and real environments is not well understood but the detailed information for the corrosiveness is vital advantage for test design and helps significantly when you need to analyse failures from the field.